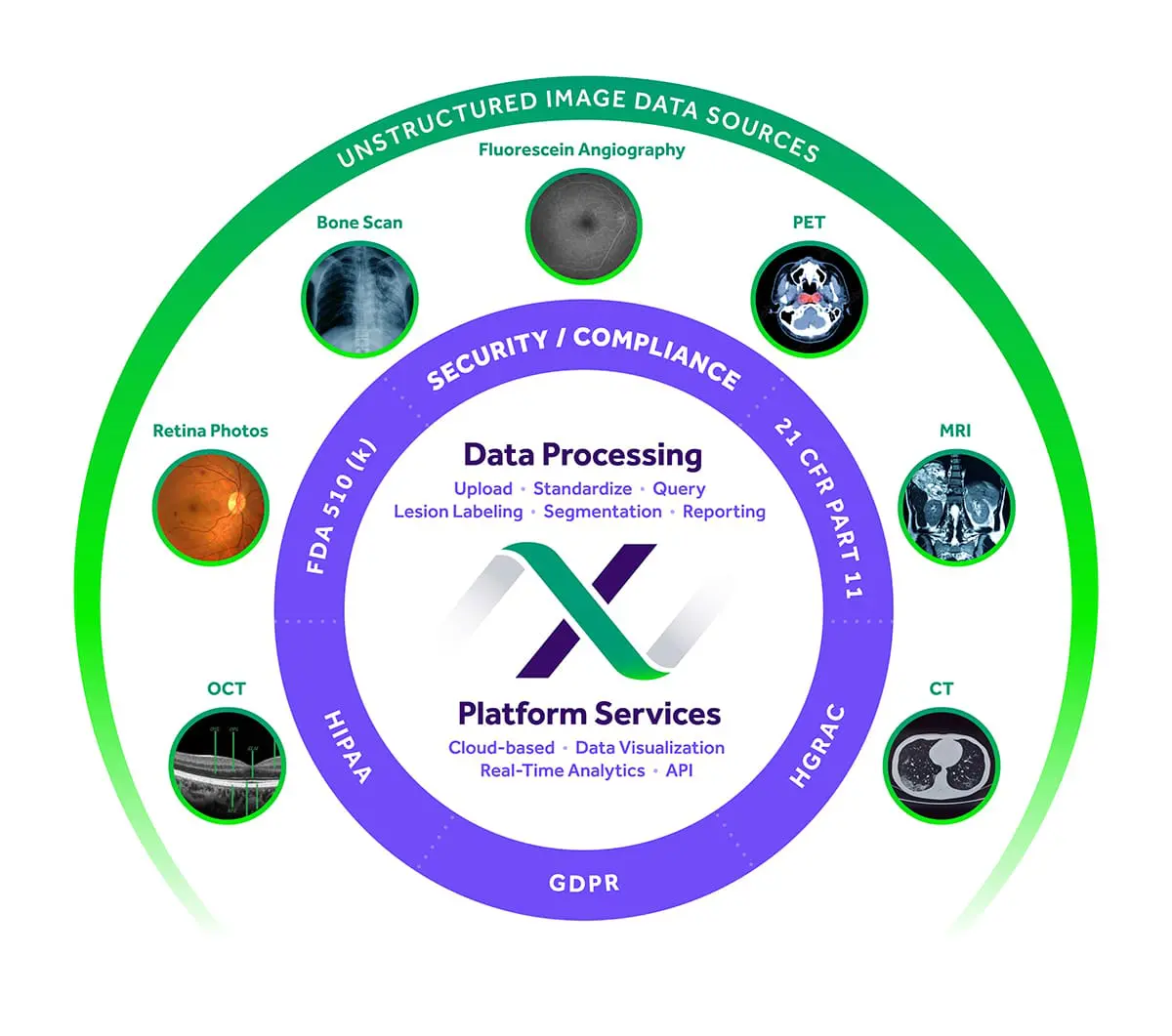
EXCELSIOR Software
MERIT’s EXCELSIORTM technology platform increases accuracy and efficiency by providing a suite of advanced endpoint analysis tools designed based on our extensive collaboration with biopharma companies.
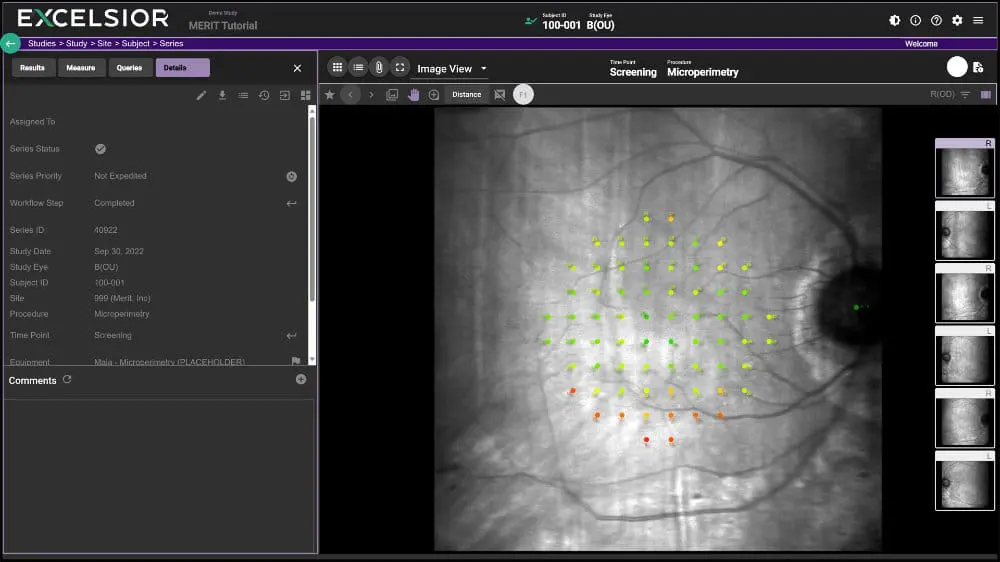
With MERIT your study data and images can easily be shared between clinical sites, reading centers, CROs, and Sponsors for maximum transparency. This makes managing global trials involving multiple reading centers and analysis of numerous modalities not only feasible, but practical.
Schedule a personalized demo of EXCELSIOR
KEY FEATURES
- CONVENIENCE: Cloud-based single platform offers quick deployment with no need to install software or configure firewalls
- CONFIGURATION: Customizable configuration provides flexibility to address the wide range of workflow and data endpoints required for each unique study
- TRANSPARENCY: The dedicated user roles, integrated query messaging system, and cloud-based service provides 24/7 real-time access and study progress transparency to all trial partners throughout the study timeline
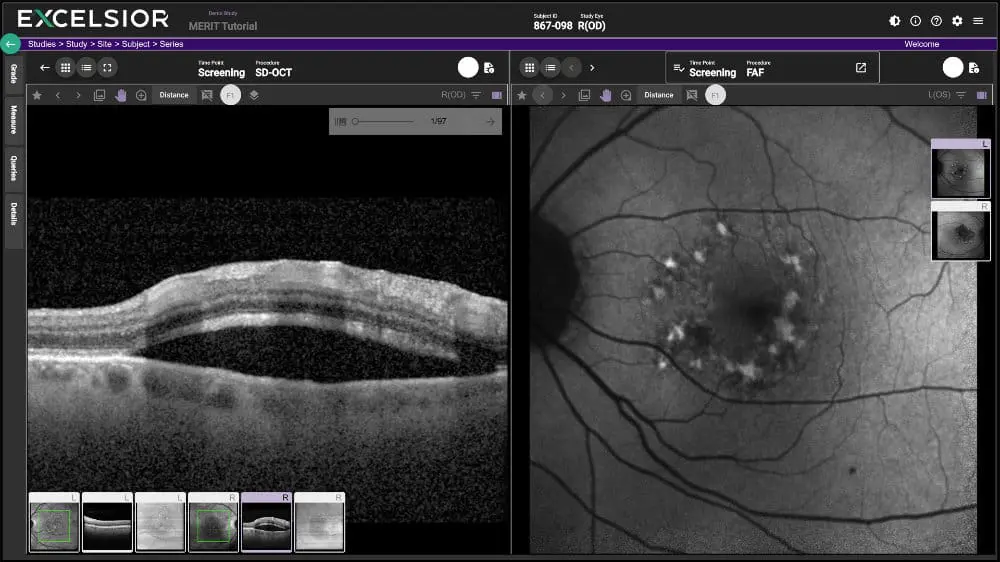
- SPLIT SCREEN: EXCELSIOR provides the ability to split a screen between two different images for different procedures which increases reader ease, efficiency, and accuracy. This comparative view can also provide the same imaging type at different timepoints, so more of a historical comparison for an individual subject and their progression using one specific procedure
- COMPLIANCE: FDA 510(k) cleared medical device for ophthalmic and radiological indications, HIPAA, 21 CFR part 11, and GDPR compliant
BENEFITS
- DATA TRANSFER: Effective and instantaneous transfer of data from one location to another. Image data can be directly and automatically moved to the sponsor’s data warehouse for further analysis
- REAL-TIME ACCESS: All stakeholders have real-time access to the curated data. Sponsors can view study progress and data in a transparent, secure environment
- NO GEOGRAPHIC LIMITATIONS: With cloud-based data infrastructure, data can be viewed and analyzed anywhere
- LATEST TECHNOLOGY: Latest web and imaging technologies for data cleaning, standardization, analysis, reading, and reporting
- EFFICIENCY: Enhanced efficiency and accuracy for data collection and interpretation. Our data acquisition platform makes site queries more efficient

Software User Testimonial
EXCELSIOR is a simple, easy to navigate system. Queries are clearly defined. MERIT’s support staff are very helpful with the initial training process and remain engaged when issuing queries or if data issues arise. In comparison to (MERIT competitor software), which we use for many other trials, I would say you have the superior product overall, especially for ease of uploads.
– Site Coordinator, EXCELSIOR User
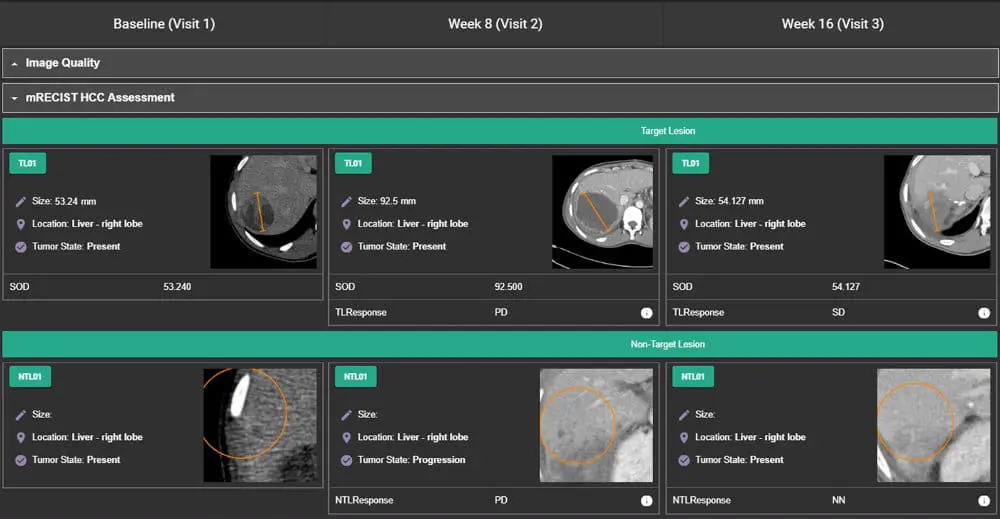
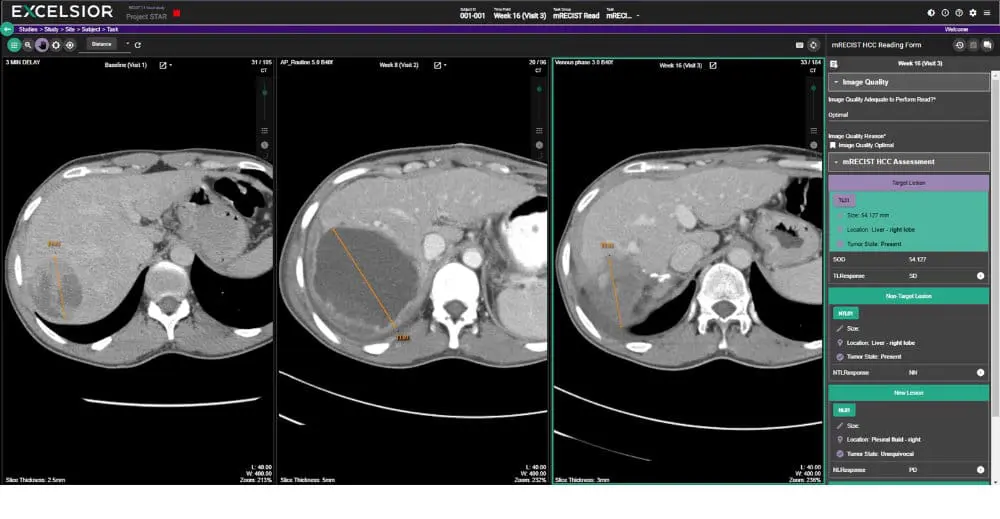
Erickson, B. J., Kirk, S., Lee, Y., Bathe, O., Kearns, M., Gerdes, C., Rieger-Christ, K., & Lemmerman, J. (2016). The Cancer Genome Atlas Liver Hepatocellular Carcinoma Collection (TCGA-LIHC) (Version 5) [Data set]. The Cancer Imaging Archive. https://doi.org/10.7937/K9/TCIA.2016.IMMQW8UQ
SPIROMETRY IN EXCELSIOR
- Scalable & flexible to adapt to trials of all phases and sizes
- Configured rapidly to collect global data in real time with secure storage
- Compatible with a wide range of equipment & technologies
- Supported by our team of in-house respiratory experts via phone or email

Spirometry
- According to 2019 ATS/ERS guidelines
- Four attempted tests, all deemed acceptable by the site