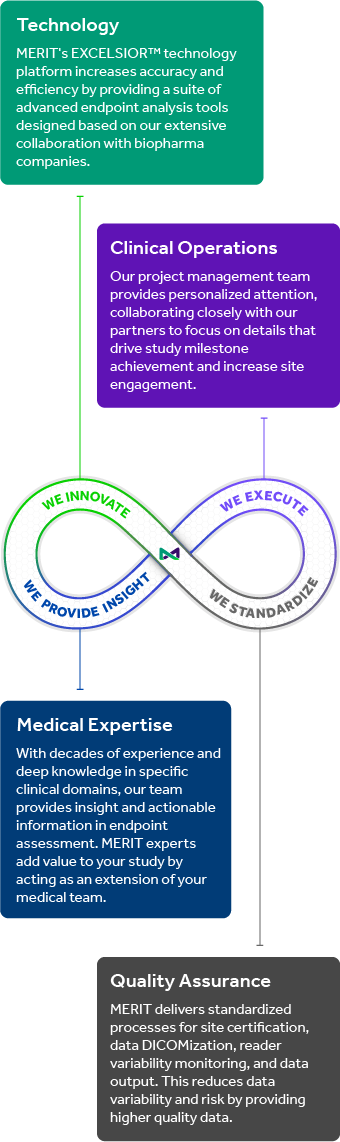
The MERIT Reading Center provides unrivaled independent reading center services built on a four-pillar framework of authoritative medical expertise, advanced FDA 510(k) cleared technology, robust global clinical operations, and our ISO-certified QMS and ISMS.
The Reading Center begins with its deep bench of Medical and Scientific Affairs leaders. They direct the activities of a rigorous, dedicated, and quality-focused team that provide masked, independent grading by carefully selected and trained readers as well as data management by highly qualified and experienced data analysts.
In addition, the Medical and Scientific Affairs team can offer insights into which combination of morphologic and visual function endpoints will best support a Sponsor’s specific target product profile and market indication.